Introduction
Materials and Methods
Plant Material
External Stimulus for Inoculation of TMV
RNA Extraction and RT-PCR
Western-blot Analysis
Purification of Recombinant Protein from Noninfected and Infected Plant Leaves
Transmission Electron Microscopy (TEM)
Results
Generation of Transgenic Plants Expressing PSA-IgG FcK
Pathogen Infection and Plant Growth of Transgenic Tobacco Expressing PSA-IgG FcK Vaccine Protein
Detection of TMV in Leaf Tissues After TMV Infection
Protein Expression Levels in Plant Leaf Tissues
No TMV in the Purified PSA-IgG FcK Samples from TMV-infected Plant Leaves
Discussion
Introduction
Recombinant therapeutic proteins, such as antibodies and vaccines, have been produced in genetically engineered tobacco plants, which are classified with horticultural plants such as tomato and petunia in the family Solanaceae. These products can be used to treat cancer, infectious diseases, chronic diseases, and other illnesses (Mohammadzadeh et al., 2014; Shoji et al., 2015;Kim et al., 2016; Kang et al., 2017). In general, these valuable immunotherapeutic proteins can be expressed and produced in mammalian, plant, yeast, insect, and bacterial expression systems (Nielsen, 2013; van Oers et al., 2015; Dumont et al., 2016; Huang et al., 2017; Mohammadinejad et al., 2019). In the plant expression system, the probability of human pathogen contamination is lower than in mammalian-derived cell systems (Moussavou et al., 2015; Lee and Ko 2017). In addition, it is easy to scale-up for mass production since plant cultivation systems require low-cost inputs such as soil, light, and water (Holtz et al., 2015; Sack et al., 2015).
Plants used for the production of therapeutic proteins are mainly Arabidopsis, tobacco, lettuce, and tomato (Zhou et al., 2008; Yanez et al., 2017; Mirzaee et al., 2018; Song et al., 2018b). Among them, many studies have been performed using the tobacco plant expression system to investigate therapeutic proteins, such as ZMApp for anti-Ebola virus antibody; Pfs25 VLP, which is a malaria vaccine candidate; and P2G12, which is an anti-human immunodeficiency virus (HIV) antibody (Floss et al., 2009; Olinger et al., 2012; Jones et al., 2013). These applied tobacco plants are easy to grow under horticultural production systems, such as a greenhouse. In addition, more biomass can be obtained from tobacco compared to other plants; moreover, tobacco has a high transformation rate. However, one of the problems with tobacco is its high risk of TMV infection.
TMV is rod shaped with a length of 300 nm and a diameter of 18 nm (Sachse et al., 2007). TMV belongs to the Tobamovirus genus of the Virgoviridae family and has a positive-sense, single-stranded RNA with 6,400 nucleotides (Alonso et al., 2013). Once the tobacco plant is infected, symptoms such as an emerging mosaic pattern, necrosis, and yellowing of plant tissue appear within 7 to 14 days (Sacristán et al., 2003; WAHYUNI et al., 2008). TMV is easily transmitted using contaminated tools and when cigarette-smoking workers rub infected leaves (Balique et al., 2012). For these reasons, there might be concerns about the use of tobacco plants grown under the horticultural greenhouse system for the production of therapeutic proteins.
In this study, prostate-specific antigen (PSA), which is commonly used as a diagnostic biomarker of prostate cancer, was expressed in the transgenic tobacco system. PSA is a representative tumor-associated antigen (TAA) that can serve as a promising vaccine candidate (Madan et al., 2006; Gulley et al., 2014). PSA protein was fused to IgG Fc fragments tagged with a KDEL endoplasmic reticulum (ER) retention signal to purify the target PSA protein efficiently using affinity chromatography. The expression levels of PSA, plant morphology, plant biomass, and purification efficacy were investigated and compared between TMV-infected and noninfected transgenic tobacco plants to determine the effect of TMV infection on the production of recombinant PSA vaccine proteins in transgenic tobacco plants.
Materials and Methods
Plant Material
Transgenic tobacco plants expressing the prostate cancer vaccine candidate, the PSA-IgG FcK protein, were used as plant materials for the TMV infection experiment. The PSA gene was fused to the immunoglobulin Fc fragment and tagged with the KDEL endoplasmic reticulum (ER) retention signal to construct the PSA-IgG FcK recombinant protein gene. The PSA-IgG FcK gene expression cassette, designed to be controlled by the constitutive 35S promoter with the untranslated leader sequence of alfalfa mosaic virus RNA 4 (AMV), was cloned into the pBI121 plant expression vector to generate the pBI PSA-IgG FcK vector. The vector was transferred into Agrobacterium tumefaciens (LBA4404) by electroporation (Song et al., 2018a). Agrobacterium-mediated plant transformation was then performed on the tobacco plant (Nicotiana tabacum) (Lu et al., 2012)
External Stimulus for Inoculation of TMV
Transgenic plants were grown in a greenhouse for 5 weeks. After dividing the plants into noninfected and infected groups (two plants, respectively), the 20 tobacco leaves for each plant were damaged by creating a cross-shaped knife scratch, and then a mashed cigarette sap solution filtered using a Miracloth (Bioscience, La Jolla, CA) was applied onto each leaf. Leaf samples were collected 3, 6, 9, 13, and 17 days after infection, and leaf morphology and infection symptoms were observed.
RNA Extraction and RT-PCR
Total RNA was extracted from leaves using TRIzol reagent (Invitrogen, Carlsbad, CA), following the TRIzol RNA isolation protocol (Song et al., 2018a). To remove genomic DNA and synthesized cDNA from the isolated RNA, the Quantitect reverse transcription kit (Qiagen, Valencia, CA) was used according to the manufacturer’s instructions. To detect TMV, each cDNA sample was used as a template, and RT-PCR was performed using the Maxime PCR premix kit (iNtRON Biotechnology, Seoul, Korea) according to the manufacturer’s instructions. PCR was performed using the following pair of primers to detect the TMV complete genome (GenBank accession No. AF273221): TMV forward primer, 5'-ACA CAC AGT TGG ACG ACG-3', and TMV reverse primer, 5'-CTC ATC AAC GAC TTC TTC TG-3'. PCR conditions were set as follows: initial denaturation at 94°C for 3 min, followed by 30 cycles of denaturation at 94°C for 30 s, annealing at 64°C for 30 s, and extension at 72°C for 45 s. Final extension was set at 72°C for 7 min. Eukaryotic translation elongation factor 1-α (EF 1-α) was used as a housekeeping gene.
Western-blot Analysis
Transgenic tobacco plant leaves were ground with 600 µL of 1X PBS (137 mM NaCl, 10 mM Na2HPO4, 2.7 mM KCl, and 2 mM KH2PO4) to extract the total soluble proteins. Each ground sample was mixed with loading buffer (1 M Tris-HCl, 50% glycerol, 10% SDS, 5% 2-mercaptoethanol, and 0.1% bromophenol blue) and loaded on a 10% SDS-PAGE gel. Proteins separated by size were transferred to a nitrocellulose membrane (Millipore Corp, Billerica, MA). The membrane was blocked with 5% skim milk (Sigma, St. Louis, MO) in TBS-T buffer [1X TBS plus 0.5% (v/v) Tween 20] for 1 h and 30 min at room temperature. The membrane was incubated with rabbit anti-PSA antibody (Abcam Inc., Cambridge, MA) as a primary antibody, after which horseradish peroxidase (HRP)-conjugated anti-rabbit IgG (H+L) (Bethyl, Montgomery, TX) was applied as a secondary antibody. HRP-conjugated goat anti-human IgG, Fcγ fragment (Jackson ImmunoResearch, West Grove, PA), was used to detect the human Fc region of PSA-IgG FcK. Chemiluminescent substrate (Bio-Rad, Hercules, CA) was used for visualizing the protein bands. Human KLK3 (Sino Biological Inc., Beijing, China) and human anti-rabies immunoglobulin (NIBSC, Hertfordshire, UK) were used as positive controls for PSA and human Fc-region, respectively. The nontransgenic leaf sample was used as a negative control. Comparison of relative band intensities was performed using ImageJ (National Institutes of Health, Bethesda, MD). Gene expression tests were performed with two biological and three technical replicates.
Purification of Recombinant Protein from Noninfected and Infected Plant Leaves
For the purification of PSA-IgG FcK, 50 g of noninfected and infected leaves were each ground in 500 mL of extraction buffer (37.5 mM Tris-HCl, pH 7.5, 50 mM NaCl, 15 mM EDTA, 75 mM sodium citrate, and 0.2% sodium thiosulfate) and were centrifuged at 8,800 × g for 30 min at 4°C. The supernatant of each sample was filtered using a Miracloth (Biosciences, La Jolla, CA), the pH was adjusted to 5.1 using acetic acid (pH 2.4), and the solution was centrifuged at 10,200 × g for 30 min at 4°C. After centrifugation, the supernatant of each sample was adjusted to pH 7.0 using 3 M Tris solution, and ammonium sulfate (Duchefa Biochemie, Haarlem, Netherlands) was added to 8% saturation. Each solution was incubated for 2 h at 4°C and centrifuged at 8,800 × g at 4°C. Ammonium sulfate was added to each supernatant to 22.6% saturation, and samples were incubated overnight at 4°C. Each solution was centrifuged at 8,800 × g for 30 min at 4°C. The pellet was resuspended in 50 mL of 1X PBS and centrifuged at 10,200 × g for 30 min at 4°C. The obtained solution was applied to rProtein A Agarose Resin (Amicogen, Jinju, Korea) to purify PSA-IgG FcK proteins according to the manufacturer’s instructions. The purified samples were confirmed by SDS-PAGE.
Transmission Electron Microscopy (TEM)
Preparation of 100 mg of TMV-infected and noninfected leaves was performed to confirm the presence of TMV particles through TEM analyses. Samples were ground in 300 µL of 1X PBS and centrifuged at 15,814 × g for 15 min at 4°C, after which the supernatant was collected. Each sample was applied to glow discharged formvar/carbon-coated grids (Ted Pella, Redding, CA) and negatively stained with 2% uranyl acetate. TMV particles were observed using a JEOL JEM-1010 transmission electron microscope (Jeol, Tokyo, Japan) operating at 80 kV.
Results
Generation of Transgenic Plants Expressing PSA-IgG FcK
The plant expression vector pBI PSA-IgG FcK was transferred into tobacco plants via Agrobacterium-mediated cocultivation (Fig. 1). Approximately 30 calluses were generated after transferring to regeneration media (Fig. 1C). Six green shoots were selected and grown in kanamycin-containing MS media. Each leaflet from six plants was sampled to confirm expression of the PSA-IgG FcK protein (‑55 kDa). Nontransgenic tobacco as a negative control had no protein band, whereas commercially available PSA antigen (‑28 kDa) and anti-rabies mAb (‑50 kDa) both showed expected protein bands (Fig. 1D). Among the six shoots, two plants (Lanes 2 and 4) showed the expected protein bands (‑68 kDa), which were doubly confirmed using the anti-PSA antibodies (left) and anti-IgG Fc (right) (Fig. 1D). For future studies, the #2 shoot with high expression levels was subcultured for multiplication and grown in a greenhouse to obtain transgenic seeds.
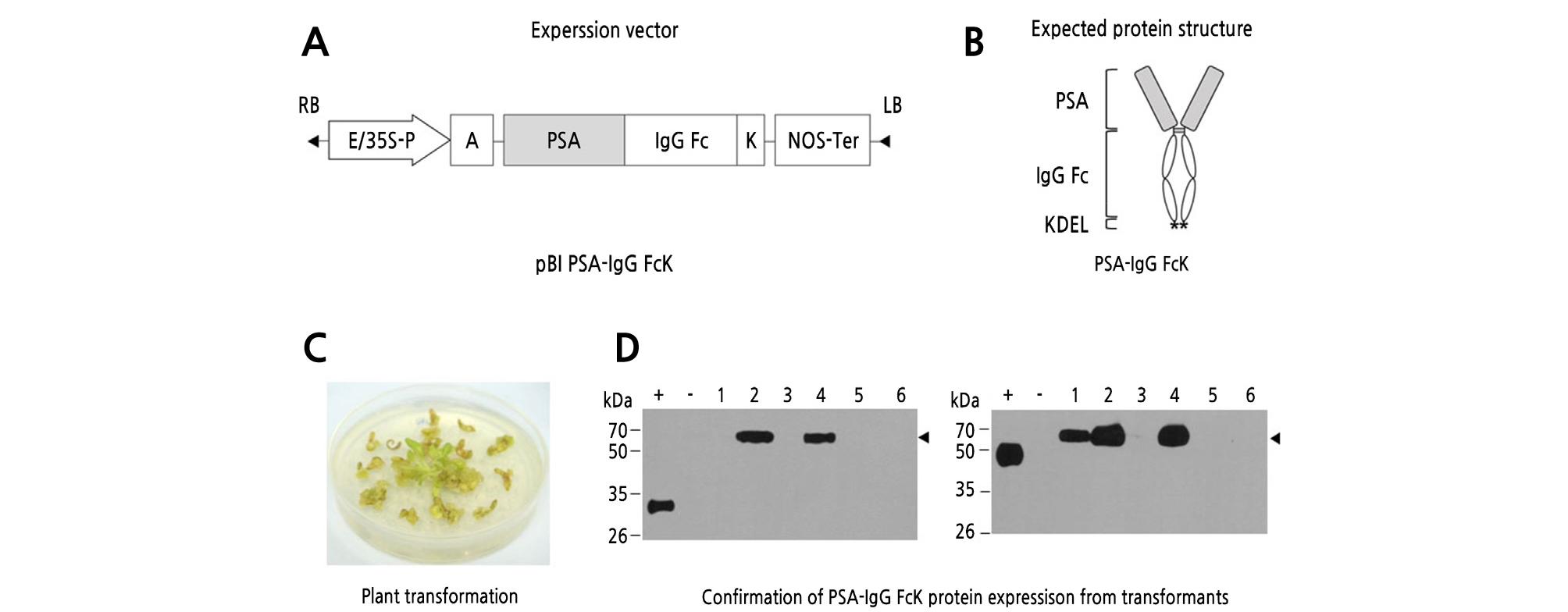
Fig. 1.
Generation of transgenic tobacco plants expressing prostate-specific antigen (PSA)-IgG FcK protein. (A) The PSA-IgG FcK gene expression cassette in the pBI121 vector. E/35S-P, cauliflower mosaic virus 35S promoter with duplicated enhancer region; A, alfalfa mosaic virus untranslated leader sequence (AMV) of RNA4; K, endoplasmic reticulum (ER) retention signal (KDEL); NOS-Ter, the nopaline synthase gene terminator. (B) Expected protein structure of recombinant PSA-IgG FcK protein: gray round square, PSA; white oval region, human IgG Fc fragment; star, KDEL. (C) Regenerants from tobacco (Nicotiana tabacum) plantlets using Agrobacterium-mediated plant transformation. (D) Western-blot analysis of PSA-IgG FcK protein expression in transformants. PSA-IgG FcK was detected with anti-PSA antibodies (left) and HRP-conjugated anti-human IgG Fc antibodies (right). Lane 1, positive control (+); lane 2, nontransgenic tobacco plant (‒); lanes 3-8, transformants.
Pathogen Infection and Plant Growth of Transgenic Tobacco Expressing PSA-IgG FcK Vaccine Protein
The leaves of 5-week-old plants raised in vivo were infected with mashed cigarette sap solution as described in Materials and Methods. The color of the leaves remained unchanged, and no symptoms developed until the third day (Fig. 2A). Phenotypic symptoms, such as circular dark yellow spots, which is typical TMV mosaic morphology, initially appeared 5 days after TMV infection. The TMV infection-related symptoms became more severe as time went by. At 17 days, several leaves became slim and were easily crushed with mild pressure. The average infected leaf numbers were 2.5, 6.5, 8.0, and 9.0, respectively (Fig. 2B). The increasing rates of virus-infected leaf numbers were the highest at around 6 ‑ 9 days compared to other time intervals (Fig. 2B).
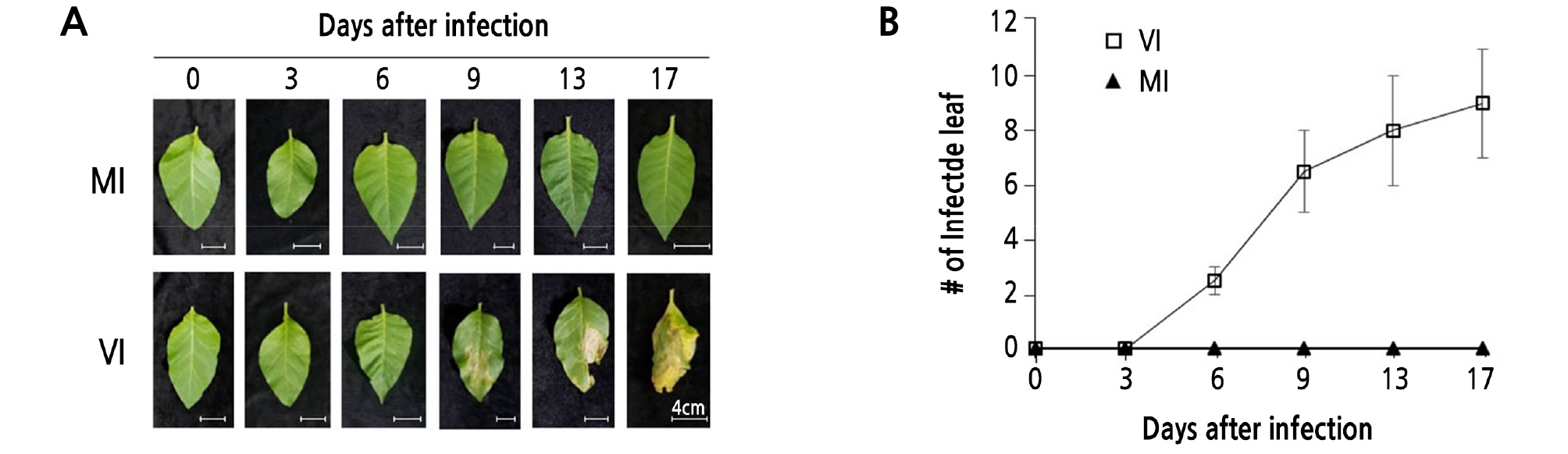
Fig. 2.
Plant leaf morphology of transgenic tobacco expressing PSA-IgG FcK vaccine protein after tobacco mosaic virus (TMV) infection. (A) Photographs were taken at 0, 3, 6, 9, 13, and 17 days after TMV infection with mashed cigarette sap solution (MA). MI, mock-inoculated transgenic plant; VI, virus-infected transgenic plant (Scale bar = 4.0 cm). (B) The number of infected leaves with typical TMV-associated mosaic patterns was counted at 0, 3, 6, 9, 13, and 17 days. Standard deviation is indicated with error bars.
Detection of TMV in Leaf Tissues After TMV Infection
After infecting plants in soil pots with TMV, the mRNA expression levels of TMV genes in the leaves were investigated. Total RNA was extracted from plants 3, 6, 9, 13, and 17 days after TMV infection. Each cDNA template was used to amplify TMV genomic fragments. The control gene EF-1α, showing the expected product size of 156 bp, was observed in all tobacco plant leaf tissue samples regardless of TMV infection (Fig. 3A). All the virus-infected leaves (days 3, 6, 9, 13, and 17) showed the expected PCR products corresponding to the TMV fragments (708 bp) (Fig. 3A). Noninfected transgenic plant leaves had no PCR product. TEM results showed that TMV (diameter 20 nm, length 250 nm) was observed in the infected leaf extract samples at 17 days (Fig. 3B). No TMV was observed in the non-TMV- infected plant leaf samples.
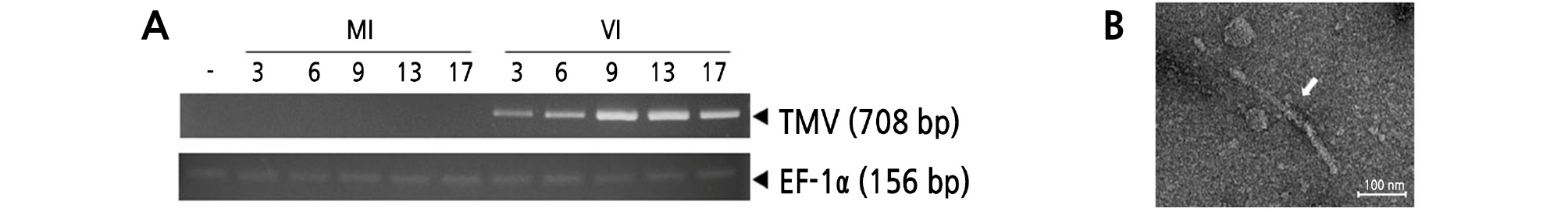
Fig. 3.
Detection of TMV in infected tobacco plant leaves. RT-PCR (A) and transmission electron microscopy (TEM) (B) analyses were conducted to confirm the existence of TMV in fresh leaf tissues with pathogenic symptoms. (A) Lane 1, wild-type tobacco; lanes 2‑ 6, MI plants sampled at 0, 3, 6, 9, 13, and 17 days; lanes 7‑ 11, VI plants sampled at 0, 3, 6, 9, 13, and 17 days. (B) TEM analysis was conducted with VI leaves sampled and 17 days, respectively. White arrow indicates TMV (Scale bar = 100.0 nm).
Protein Expression Levels in Plant Leaf Tissues
Western-blot analysis was conducted to investigate the effect of TMV infection on PSA-IgG FcK protein expression level (Fig. 4). The middle portions of the tobacco plant leaves [mock inoculated (MI) and virus infected (VI)] were sampled to extract total soluble proteins. The protein expression levels of PSA-IgG FcK did not differ significantly between the MI and VI plants. The relative protein expression levels were confirmed by anti-PSA and anti-IgG Fc antibodies, respectively (Fig. 4A left and right). The density of the 55-kDa protein band was calculated using ImageJ (NIH, Bethesda, MD) (Fig. 4B left and right). TMV-infected leaf samples at 17 days showed the lowest band intensity compared to the other experimental groups (Fig. 4A). In both MI and VI plants, similar variation of the protein band was observed 17 days after TMV infection. These results suggested that TMV infection did not significantly affect protein expression levels.
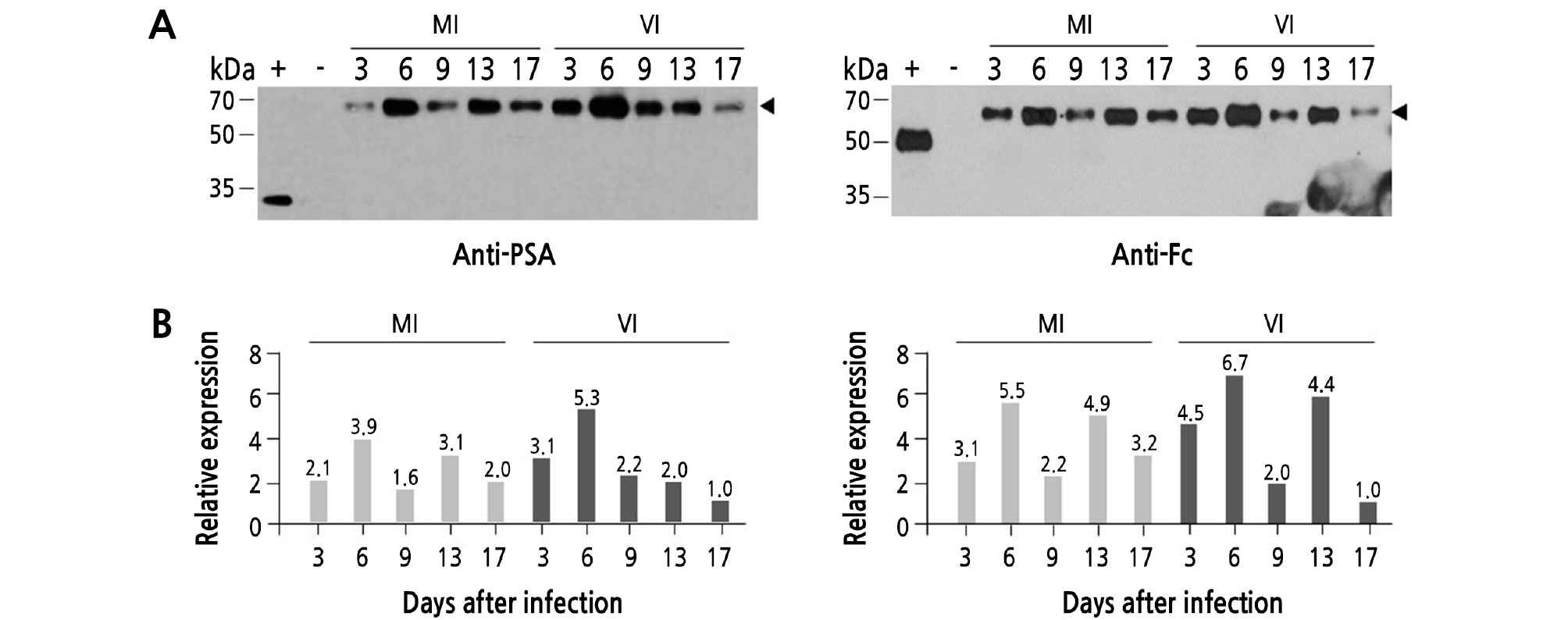
Fig. 4.
Protein expression levels of PSA-IgG FcK vaccine protein in transgenic tobacco leaves after TMV infection. To determine protein expression levels of each experimental group (MI and VI), Western-blot analysis was performed. (A) The PSA-IgG FcK vaccine protein expression was double-checked using the anti-PSA antibody as a primary antibody and then incubated with HRP-conjugated anti-rabbit IgG (H+L) as a secondary antibody for PSA (left) and HRP-conjugated anti-human IgG Fcγ fragment-specific antibody for IgG Fc fragment (right). +, Commercialized PSA protein (A, left), human anti-rabies immunoglobulin (A, right); -, wild-type tobacco (A right and left); lanes 3‑ 7, MI plants sampled at 0, 3, 6, 9, 13, and 17 days (A right and left); lanes 8‑ 12, VI plants sampled at 0, 3, 6, 9, 13, and 17 days (A right and left). (B) Relative band density of PSA-IgG FcK quantified using ImageJ software (NIH, Bethesda, MD) to analyze the X-ray film image. The relative value of the expression rate is indicated on the top of the bar in the graph.
No TMV in the Purified PSA-IgG FcK Samples from TMV-infected Plant Leaves
Approximately 50 grams of both MI and VI transgenic tobacco leaves was harvested on day 17 after infection to purify PSA-IgG FcK fusion vaccine proteins. The proteins were efficiently captured and mainly eluted in fraction #1. The strongest protein bands were mainly present in fraction #1 of both MI and VI. The total amount of PSA-IgG FcK fusion vaccine protein obtained from infected plants (VI) was similar to noninfected plants (MI). SDS-PAGE showed that the molecular weight of the target protein was approximately 55 kDa, and the purity of the PSA-IgG FcK fusion vaccine protein was not significantly different between noninfected and infected plants (Fig. 5A). The western-blot showed bands that were approximately 55 kDa using both anti-PSA and anti-Fc antibodies (Fig. 5A and 5B, respectively).
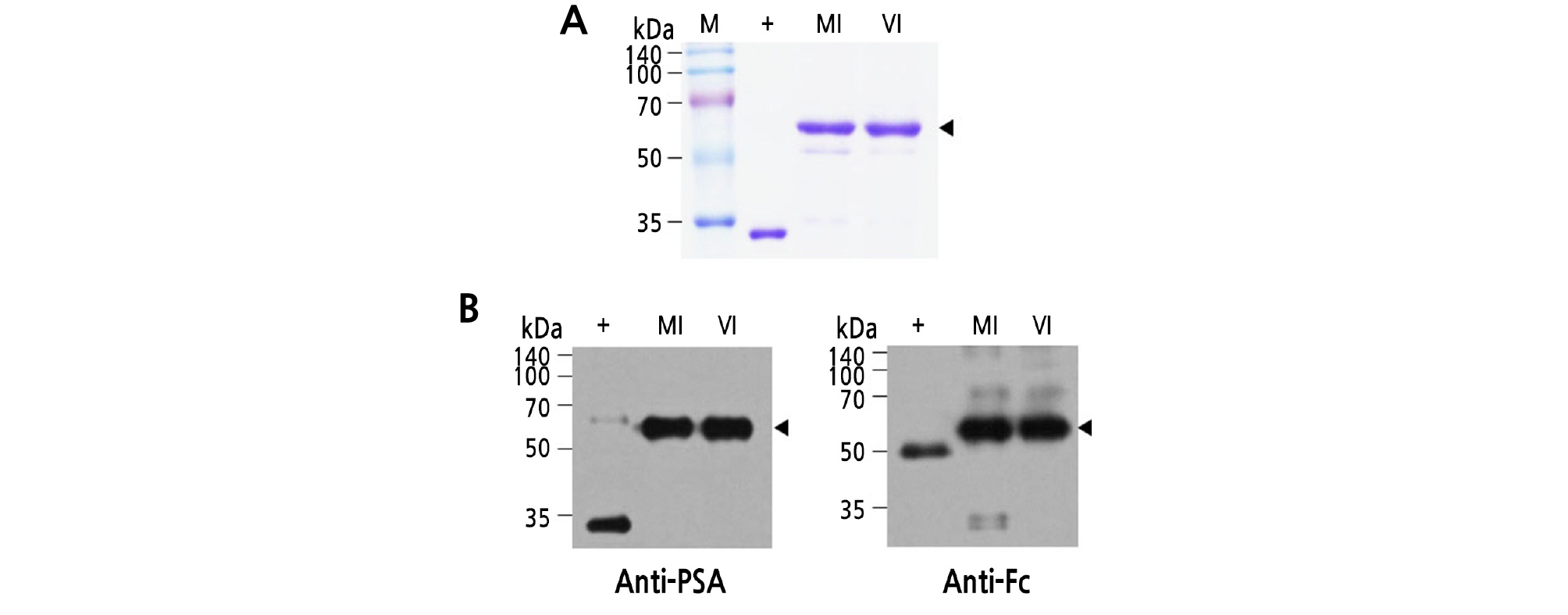
Fig. 5.
Purification of PSA-IgG FcK fusion vaccine protein from MI and VI plant leaf biomass. (A) SDS-PAGE analysis of the eluted F1 fraction samples obtained from each experimental group (MI and VI). Samples (20 µL) were loaded in each well. Lane 1 (M), protein marker; lane 2 (+), human PSA protein as a positive control (‑28 kDa); lane 3, purified PSA-IgG FcK from a MI plant; lane 4, PSA-IgG FcK purified from a VI plant. (B) Western-blot analysis was performed to reconfirm expression of the target recombinant vaccine protein using the two methods described in Fig. 4. +, Commercialized PSA protein (left), human anti-rabies immunoglobulin (right); lane 2, PSA-IgG FcK purified from MI plant; lane 3, PSA-IgG FcK purified from VI plant.
Discussion
The main goal of this study was to confirm the effect of TMV infection on phenotype and the expression of therapeutic recombinant proteins in transgenic tobacco plants. In plant molecular biofarming, healthy plant growth conditions are one of the most important factors to ensure that plant biomass cost-effectively produces immunotherapeutic recombinant proteins such as vaccines and antibodies. However, plant pathogens such as viruses, bacteria, and nematodes can negatively affect plant biomass growth, which consequently decreases the productivity of the recombinant proteins. In this study, transgenic tobacco expressing recombinant PSA-IgG FcK vaccine candidate protein was obtained through Agrobacterium-mediated transformation. Transformants with high expression levels were selected using kanamycin- containing MS medium and western-blots. The PSA-IgG Fc was fused to a KDEL motif to increase protein expression levels and reduce plant-specific glycan residues (Kim et al., 2018; Song et al., 2018a, Song et al., 2018b; Shin et al., 2019).
Among the preliminary research to select infecting methods (Suppl. Fig. 1s), MA application was observed to be the most effective method for infecting leaves with TMV. As such, MA was applied to 5 week-old in vivo-cultured plant leaves that were scratched with a knife to create a cross-shaped wound. Typical TMV symptoms emerged on the leaves of theplants after 5 days, and the yellow spots were observed to increase as time passed. It is speculated that TMV propagation to adjacent leaf areas was accelerated by cell-to-cell movement (Ueki et al., 2010; Peiró et al., 2014).
TMV gene transcription levels tended to increase over time after infection, showing that TMV increasingly proliferated as time passed. All mechanically infected leaves showed TMV genes in the tissue regardless of the extent of pathogenic symptoms, similar to what was reported in previous studies (Torok and Randles, 2001; Iftikhar et al., 2015). However, therapeutic vaccine PSA-IgG FcK expression levels were not significantly different between MI plants and VI plants. These results indicated that TMV infection did not significantly affect the recombinant protein expression in transgenic tobacco plants.
TMV has been used as a virus-based vector to produce valuable recombinant proteins in transient plant expression systems (Jin et al., 2015; Ma et al., 2019). However, the effect of TMV infection on expression of recombinant proteins in transgenic plants has not reported yet.
In addition, we hypothesized that TMV viral particles would be removed through a purification process for the target therapeutic proteins. SDS-PAGE analysis showed that the purity of the recombinant PSA-IgG FcK derived from both MI and VI plants was not significantly different. Furthermore, there was no TMV in the purified proteins obtained from the VI plant (Suppl. Fig. 2s). It is speculated that TMV particles were efficiently removed by the purification process.
In conclusion, both healthy plants and TMV-infected plants grown in a horticultural containment greenhouse facility can be harvested and utilized to obtain recombinant vaccine proteins regardless of TMV-induced pathogenic symptoms. This research is meaningful with respect to tobacco cultivation for plant molecular biofarming.
Supplementary Material
Supplementary materials are available at Horticultural Science and Technology website (https://www.hst-j.org).
- HORT_20200027_Fig_1s.pdf
The phenotype of in vitro tobacco plant leaves with four different tobacco mosaic virus (TMV) infection external stimuli. Prior to the main infection experiment, a preliminary experiment was conducted to establish the virus infection route in tobacco plants. To accelerate virus infection, plant leaves were lightly scratched using a knife to create a cross-shape (bottom panel) and these leaves were compared to the non-cut group (upper panel). Four treatments were conducted for each experimental group (HT, hand touch; SA, saliva; SM, smoke; MA, mashed cigarette sap solution). Photographs were taken six days after pathogen inoculation. Among the four different external stimuli, the experimental group in which MA was applied onto the scratched leaf tissues showed the most severe symptoms among the leaves of plants with high expression levels, grown under in vitro conditions, and transplanted into a soil pot in a greenhouse.
- HORT_20200027_Fig_2s.pdf
Detection of TMV in purified samples derived from each MI (left) and VI (right) transgenic plant leaf. Purified samples were dialyzed with 1X PBS, and uranyl acetate was used as a negative stain. TEM analysis was conducted to confirm the existence of TMV in each purified elution (Scale bar = 100.0 nm).




