Introduction
Materials and Methods
Plant Material
Chemicals
Sample Preparation
Determination of Total Phenolic Content
Determination of Total Flavonoid Content
Antioxidant Activities Determined by the DPPH and ABTS Methods
HPLC Analysis of Phenolic Compounds
Determination of Amino Acids
Statistical Analysis
Results and Discussion
Antioxidant Capacities and Their Correlations with Bioactive Compounds
Free Amino Acid Composition
Conclusion
Introduction
Asparagus (Asparagus officinalis L.) is a perennial crop with edible, young shoots, commonly called spears. Asparagus is a highly valued specialty crop produced in early spring to autumn in South Korea. Asparagus contains a large amount of antioxidants and other health-promoting compounds and is grown commercially worldwide, with two main types of spears produced (green and white) (Kang et al., 2017). The cultivation area of asparagus in South Korea is about 60 ha as of 2016, which is expected to increase to 140 ha in 2020, and production is expected to increase by 4000 tons (Yoo et al., 2017; Kang et al., 2018). Green spears emerge from the soil in the presence of sunlight. In contrast, white spears are grown in the dark, usually covered with sandy ridges or opaque plastic film (Jishi et al., 2012; Siomos, 2018). Asparagus is divided into American varieties and European varieties. Approximately 50 varieties of asparagus were cultivated for commercial purposes around the world in 2017. Asparagus varieties vary in productivity and quality characteristics, and they include 100% male or male and female varieties. The merits of male varieties include high productivity, with yields of up to 20% higher than those of male and female varieties, but not in all varieties (Siomos, 2018). Male varieties are generally resistant to diseases such as Fusarium. In particular, the European Limgroup B.V. varieties are all 100% male varieties (Kang et al., 2017).
The variety mainly cultivated in South Korea is North American Atlas. Atlas spears are green and medium to large in size with a light purple color at the apical part and under the petiole. This variety grows in a solid, upright form under high-temperature growth conditions (Kang et al., 2017). It is resistant to Fusarium, rust, and Cercospora blight as well as asparagus virus (AV) II.
Asparagus contains a significant bioactive compound content, which is potentially a major health benefit. There is a good correlation between the total phenolic content, total flavonoid content, and antioxidant capacity (2,2-diphenyl- 1-picrylhydrazyl [DPPH] and 2,2'-azinobis-3-ethylbenzothiazolin-6-sulfonic acid [ABTS] scavenging activity) in some Malvaceae species, lemon, and other vegetables (Vinson et al., 1998; de Oliveira et al., 2012; Xi et al., 2017). The most useful components in asparagus are rutin, a flavonoid derivative of the flavonol aglycone quercetin; protodioscin, a furostanol saponin; and glutathione, a biothiol (Motoki et al., 2012; Drinkwater et al., 2014; Siomos, 2018). Environmental conditions have a significant effect on the antioxidant compound content in asparagus spears (Jishi et al., 2012; Ku et al., 2018). Rutin is the major determinant of antioxidant activity in green asparagus (Motoki et al., 2012). Antioxidants are key substances that protect cells and tissues by minimizing oxidative stress caused by free radicals (Yoon et al., 2019). Protodioscin has been associated with bioactivities, including cytotoxicity against several human cancer cell lines (Wang et al., 2003). This compound is more abundant in white asparagus than in green asparagus, mainly in the bottom part.
In this study, we investigated the major bioactive compounds in seedlings of 23 varieties of asparagus and investigated the correlation between these components and antioxidant capacity. The distribution of free amino acids in each variety was confirmed. Based on the results, we will provide basic data for spear production by comparing the useful components in seedlings of 23 asparagus varieties.
Materials and Methods
Plant Material
Twenty-three varieties of asparagus seeds were purchased from various seed companies (Table 1) and grown in the tissue culture room of Gangneung-Wonju National University in Gangneung, South Korea (Table 1). Asparagus seeds were dipped in 70% ethanol for 1 min, immersed in a 2% sodium hypochlorite solution, and gently shaken for 10 min to sterilize the seeds. Thereafter, the cells were washed three times with sterilized water and then soaked in water for 5 h. The soaked seeds were then drained and cultured in petri dishes containing 25 mL of Murashige and Skoog (MS) solid medium. The culture conditions were maintained at 25°C under incandescent light for 16 h. The incubation period was 1 month. The length of the seedlings grown for 1 month was approximately 10 cm. All biological activity analysis was performed using plant parts except roots.
Table 1. Asparagus varieties and origin
Chemicals
All chemicals used, including the solvents, were of analytical grade. DPPH free radical (90% purity), ABTS, Folin- Ciocalteu’s phenol reagent, sodium carbonate, rutin (94% purity, high-performance liquid chromatography [HPLC] grade), and kaempferol 3-O-rutinoside (98% purity, HPLC grade) were purchased from Sigma-Aldrich (St. Louis, MO, USA). The other solvents used in the HPLC analysis were purchased from Daejung Chemicals (Siheung, Korea). The buffer used for free amino acid analysis included RH-1, RH-2, RH-3, RH-4, and RH-RG from Kanto Chemical Company (Tokyo, Japan). Ninhydrin reagent, buffer solution, and each individual amino acid standard were purchased from Wako Pure Chemical (Osaka, Japan).
Sample Preparation
Growing seedlings of the asparagus varieties were collected and stored frozen in sealed, clear polyethylene plastic bags at ‑ 80°C for later use. Samples grown under the same conditions were freeze-dried and ground into a fine powder using a mortar and pestle. The powdery samples (100 mg) were extracted with 1 mL of 70% ethanol for 12 hours at 30°C in a water bath. After centrifugation at 26,712 g for 15 min, the supernatant was filtered through a 0.22-µm syringe filter and used for analysis.
Determination of Total Phenolic Content
Total phenolic compounds were determined according to the Folin-Ciocalteu reaction method, using gallic acid as a standard (Anesini et al., 2008). The extracts from seedlings of 23 asparagus varieties were diluted to 10,000 µg·mL-1 with 70% ethanol. An aliquot of 100 µL of each extract was mixed using a vortexer with 50 µL of Folin-Ciocalteu reagent for 3 min. Then, 300 µL of a 20% Na2CO3 solution was added, and the samples were reacted for 15 min at room temperature. The absorbance was measured at 738 nm with the microplate photometer (Multiskan FC, Thermo Scientific, MA, USA), and a calibration curve (R2 = 0.9964) was generated using gallic acid. The results were expressed as mg gallic acid equivalents (GAE)·g-1 dry weight (DW).
Determination of Total Flavonoid Content
Total flavonoids were determined according to the modified method of Re et al. (1999). Seedling extracts of 23 asparagus varieties were diluted to 10,000 µg·mL-1 with 70% ethanol. A total of 500 µL of each extract was mixed with 100 µL of 10% aluminum nitrate and 100 µL of 1 M potassium acetate. After a 40-min reaction at room temperature, the absorbance was measured at 405 nm with the microplate photometer (Multiskan FC, Thermo Scientific, MA, USA). A calibration curve (R2 = 0.9998) was generated using quercetin, and the results were expressed as mg quercetin equivalents (QE)·g-1 DW.
Antioxidant Activities Determined by the DPPH and ABTS Methods
To determine the antioxidant activity, seedling extracts of 23 asparagus varieties were diluted to 10,000, 5,000, and 2,500 µg·mL-1 with 70% ethanol. The DPPH radical scavenging assay was performed using the modified method of Re et al. (1999). A mixture of 100 µL of DPPH ethanol solution (0.15 mM) was added to an equal volume of sample extracts. After a 30-min reaction in the dark, the absorbance was measured at 517 nm. Ascorbic acid was used as a standard.
The ABTS radical scavenging activity assay was conducted according to Ku et al. (2018) with modifications. ABTS solution was dissolved in distilled water to a concentration of 7.4 mM and mixed with 2.45 mM potassium persulfate in a 1:1 ratio to produce the ABTS radical cation (ABTS+). This solution reacted overnight and was diluted with phosphate- buffered saline (PBS), and the absorbance was measured at 738 nm with the microplate photometer (Multiskan FC, Thermo Scientific, MA, USA). Ascorbic acid was used as a standard.
HPLC Analysis of Phenolic Compounds
Analysis of phenolic compounds was conducted on a Prominence HPLC system (Shimadzu, Kyoto, Japan) equipped with a diode array UV-Vis detector for monitoring at 330 nm. Compounds were separated using a C18 column (250 × 4.6 mm, 5 µm, Shimadzu, Kyoto, Japan). Binary gradient elution was performed with solvent A (S)-9-fluoro-3-methyl- 7-oxo-10-(piperazin-1-yl)-3,7-dihydro-2H-[1,4]oxazino[2,3,4-ij]quinoline-6-carboxylic acid (3DW) with 0.1% formic acid) and solvent B (acetonitrile with 0.1% formic acid), which were delivered at a flow rate of 0.7 mL/min as follows: 0 min, 12% B; 20 min, 30% B; 50 min, 80% B; 53 min, 80% B; 54 min, 88% B; and 60 min, 88% B. The injection volume was 10 µL, and the column temperature was 40°C.
Determination of Amino Acids
Free amino acids were analyzed on an amino acid analyzer (Model L-8800, Hitachi Co., Tokyo, Japan) based on the ninhydrin method. Approximately 1 g of sample was taken, and hydrolysis was carried out using 5 mL of 6 N HCl at 110°C for 22 hours. Filtered hydrolysate was dried in a vacuum evaporator and then dissolved using sonication with 10 mL of triple-distilled water. One microliter of the solution was injected directly into the amino acid analyzer. Analytical #2622 (4.6 mm × 60 mm) and guard #2650 (4.6 mm × 40 mm) columns were used for free amino acid determination. Immediately after injection into the columns, an autosampler was used for the inline derivatization by ninhydrin postcolumn derivatization. The ninhydrin-derivatized amino acids were monitored at 570 and 440 nm.
Statistical Analysis
All analyses were repeated in triplicate, and the results were statistically evaluated using a simple ANOVA with a 95 % confidence interval in SAS (version 9.1). The significance of each measurement was analyzed by Duncan’s multiple range test (DMRT) with a p ≤ 0.05 significance level. To investigate the relationship between groups, we visualized data using R (version 3.3.1) and performed heat map (ggplot2, https://cran.rstudio.com/src/contrib/ggplot2_3.1.0.tar.gz) and correlation analysis (PerformanceAnalytics, https://cran.rstudio.com/bin/windows/contrib/3.3 /PerformanceAnalytics_ 1.5.2.zip; corrplot, https://cran.rstudio.com/bin/windows/contrib/3.3/corrplot_0.84.zip).
Results and Discussion
Antioxidant Capacities and Their Correlations with Bioactive Compounds
In this study, a hierarchical clustering analysis (HCA) was performed to determine the composition of antioxidant capacities, total phenolic content, total flavonoids content, rutin, and nicotiflorin. The results of HCA are shown in the form of a heat map in which the relative concentration values for each factor are in the color range. According to the HCA results, the relative characteristics of physiological activity of 23 asparagus seedling varieties were not affected by the region of origin (Table 1). The data are classified into four clusters and show high physiological activity in the ‘HEM’, ‘TEM’, ‘MAM’, ‘POM’, and ‘UC7’ varieties (Fig. 1).
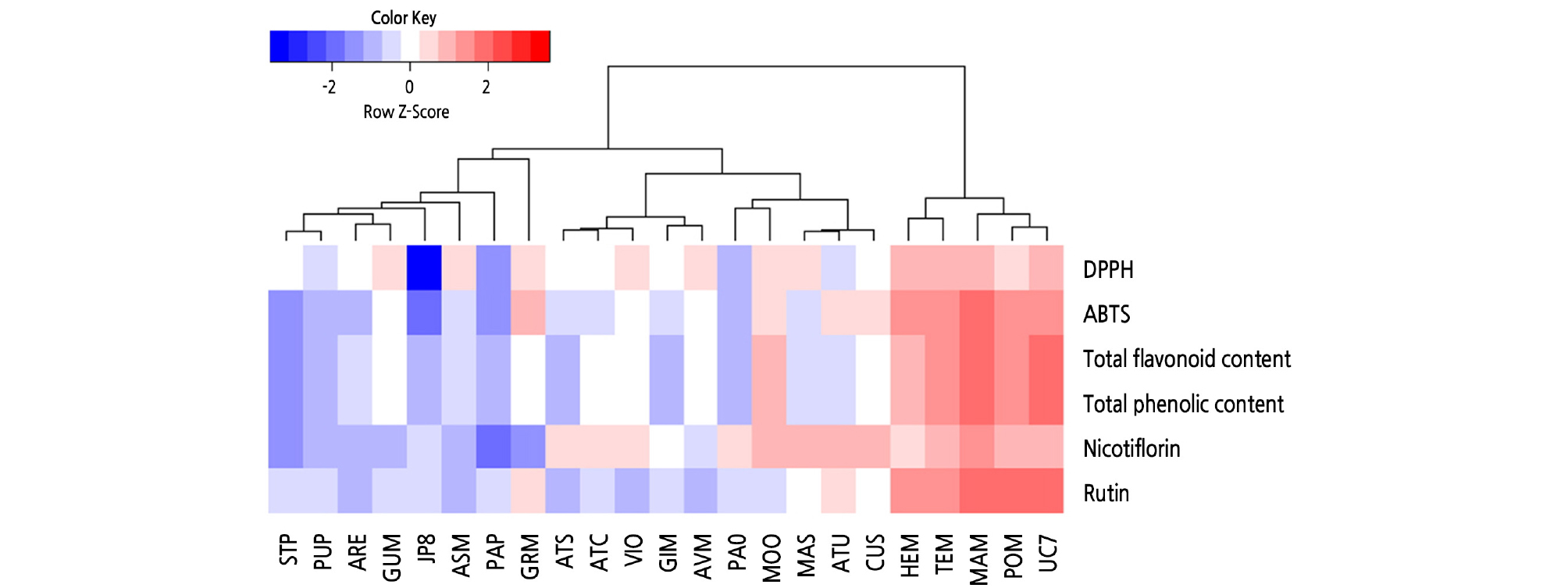
Fig. 1.
Heat map representing differences of antioxidant activities (DPPH and ABTS), total phenolic content, total flavonoid content, nicotiflorin, and rutin in 23 asparagus seedling varieties. These data were standardized. Red or blue indicates that the amino acid content is increased or decreased, respectively.
‘MAM’ contained the highest total phenolic content, which was approximately 2,044 mg GAE/g DW, followed by ‘UC7’, ‘HEM’, ‘TEM’, ‘POM’, and ‘MOO’, which contained more than 1,500 mg GAE/g DW (Suppl. Table 1s). The content of other varieties ranged widely from 874 to 1,255 mg GAE/g DW. The lowest total phenolic content was quantified in ‘JP8’ and especially ‘PUP’ and ‘STP’ in this study. The ‘PAP’, ‘PUP’, and ‘STP’ varieties also produced purple seedlings.
The varieties with a higher total flavonoid content were similar to those with a higher total phenolic content, except for ‘GRM’. The total flavonoid content of ‘MAM’ was 190 mg QE/g DW, which was the highest among all varieties and approximately 10 times higher than that of the ‘JP8’, which exhibited the lowest content. The total flavonoid content was more than 107 mg QE/g DW in ‘MAM’, ‘UC7’, ‘HEM’, ‘POM’, ‘TEM’, and ‘GRM’ (in descending order), while the other varieties contained less than 50 mg QE/g DW. Similar to the result for total phenolic content, there was wide diversity in the total flavonoid content among the varieties. The purple varieties exhibited lower phenolic and flavonoid contents.
Antioxidant capacity is required to remove the reactive oxygen species (ROS) produced by plants. It is essential that vegetables have a high antioxidant capacity to cope with ROS (Rodríguez et al., 2005; Govindan and Muthukrishnan, 2013; Wang et al., 2017). DPPH and ABTS assays are widely used to screen for antioxidant capacity in fruits and vegetables. The major difference between the DPPH and ABTS radical scavenging activity assays is that the DPPH assay can identify only hydrophilic antioxidants, while the ABTS assay can measure both the hydrophilic and hydrophobic antioxidant activity present in the sample (Arnao, 2000). The DPPH and ABTS radical scavenging activity, which is expressed in terms of the half maximal inhibitory concentration (IC50), is shown in Suppl. Table 2s relative to the known value for the antioxidant L-ascorbic acid. A lower IC50 value indicates more antioxidant potential. The DPPH assay is based on scavenging the free radical and reducing it to 2,2-diphenyl-1-picrylhydrazine, which is accompanied by a change in color from violet to yellow (Rajurkar and Hande, 2011; Govindan and Muthukrishnan, 2013). The scavenging effects on DPPH in the asparagus seedlings were excellent, especially in ‘MAM’, ‘UC7’, ‘HEM’, ‘POM’, ‘TEM’, ‘GRM’, and ‘MOO’. ‘MAM’ exhibited the best antioxidant properties (significantly lower IC50 value = 0.78; p ≤ 0.05), and ‘JP8’ possessed very poor antioxidant activity (significantly higher IC50 value = 5.25; p ≤ 0.05). A stable and colored radical cation (ABTS•+) shows a maximum absorbance at 734 nm and a decrease with the scavenging of proton radicals. Antioxidant compounds in the vegetable and fruit quench the color and produce discoloration of the preformed radical solution that is proportional to their amount (Rajurkar and Hande, 2011). A higher capacity for ABTS scavenging was expressed as a lower IC50 value. High antioxidant activity (significantly lower IC50 value = 0.59; p ≤ 0.05) was measured in ‘MAM’ in the DPPH assay, followed by ‘UC7’, ‘HEM’, ‘POM’, ‘TEM’, and ‘GRM’. The results for ABTS were similar to those of the DPPH assay. Additionally, the lowest antioxidant activity (significantly higher IC50 value = 1.82; p ≤ 0.05) was observed in ‘JP8’.
The main phenolic compounds present in asparagus are flavonoids. Three representative flavonol glycosides are rutin (quercetin-3-O-rutinoside), nicotiflorin (kaempferol-3-O-rutinoside), and narcissin (isorhamnetin- and isorhamnetin- 3-O-glucoside) (Fuentes-Alventosa et al., 2008). Rutin is predominant in asparagus spears (Motoki et al., 2012), while in the seedling stage, rutin and nicotiflorin are predominant, and some chlorogenic acid is present in small amounts. In this study, rutin and nicotiflorin were measured (Suppl. Table 3s). Similar to the pattern observed for the spear portion of asparagus, the rutin content of 16 asparagus varieties was higher in the seedling stage. The variety containing the largest amount of rutin was ‘MAM’, in which the content was 1,624 µg·g-1 DW, followed by ‘POM’, ‘UC7’, ‘HEM’, and ‘TEM’. The rutin content was significantly different among the varieties. The rutin content of ‘ASM’, ‘AVM’, ‘VIO’, and ‘ARE’ was 10 times lower than the maximum content, i.e., less than 52 µg·g-1 DW. In general, the main flavonoid in asparagus spears is rutin, and nicotiflorin is present at less than 5 mg·g-1 or is almost undetectable (Fuentes-Alventosa et al., 2008). However, at the seedling stage in asparagus, rutin and nicotiflorin were detected at the same time, and the nicotiflorin content was also high. The nicotiflorin content differs among varieties, but there is a relatively small amount relative to the rutin content. However, for some varieties (‘ATC’, ‘ATS’, ‘AVM’, ‘MOO’, and ‘VIO’), there is more nicotiflorin than rutin, and the ‘VIO’ in particular contains more than twice as much nicotiflorin as rutin (Suppl. Table 3s). The amount of nicotiflorin, which was high in the seedling stage, was markedly lower in spears (Fuentes-Alventosa et al., 2008).
Flavonoids are compounds derived from plants that are widely distributed in fruits and vegetables. Flavonoids have beneficial effects in protecting the body from various diseases such as cancer and cardiovascular disease (Panche et al., 2016). These physiological benefits of flavonoids are generally associated with antioxidant capacity and the ability to eliminate free radicals. Thus, flavonoids have the potential to cure various diseases. Studies have also shown that flavonoids such as rutin and nicotiflorin have the ability to protect neurons under stress conditions such as hypoxia and oxidative stress (Morrone et al., 2015). The rutin content in plants is highly dependent on light, and UV-B radiation has an important influence on rutin synthesis (Motoki et al., 2012). Additionally, the rutin content in asparagus is higher in green asparagus than in white asparagus and higher in the top part (6.7 mg·g-1 DW) than in the bottom part (1.9 mg·g-1 DW). This pattern occurs because the synthesis of rutin depends on light, so rutin production in the top part is high. In this study, rutin synthesis continued because the seeds of asparagus were germinated and continually exposed to light until they reached the seedling stage. Additionally, the spear thickness and rutin content of green asparagus were inversely proportional, and the rutin content decreased as the diameter increased (Stoffyn et al., 2012). The accumulation of rutin is generally distributed in epidermal tissue.
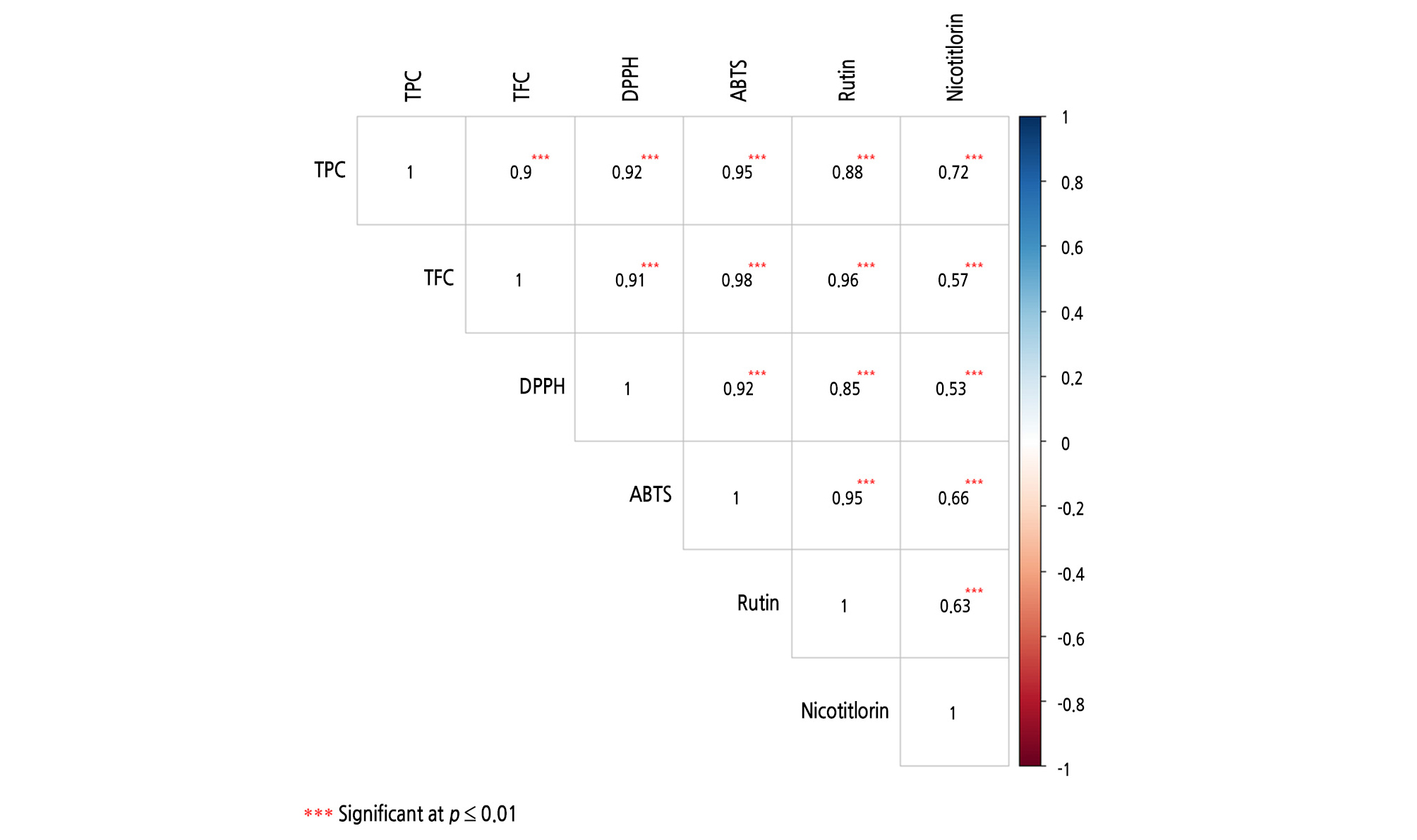
Previous studies have shown a strong correlation between antioxidant capacity and total phenol content in asparagus spears (Rodríguez et al., 2005). In particular, the major phenolic compound present in asparagus seedlings was flavonoids. In this study, the antioxidant capacity (DPPH and ABTS) and flavonoids in asparagus seedlings were strongly correlated (R = 0.91 and 0.98, respectively) (Fig. 2).
Rutin, a type of flavonoid, was shown to have a significant positive relationship with DPPH and ABTS (R = 0.85 and 0.95, respectively). However, nicotiflorin exhibited a relatively low R value with DPPH and ABTS but was highly significant based on testing (R = 0.53 and 0.66; p ≤ 0.01).
The quality of the perennial crop asparagus is influenced not only by the current cultivation environment but also by the cultivation environment of the previous year. Even if the same variety is cultivated, the total phenolic compound content may double in response to the environment. In addition, total phenolic compounds are strongly affected by genetics and directly related to antioxidant activity, which is a major factor in choosing a variety (McCormick and Thomsen, 1990; Guillén et al., 2008; Fanasca et al., 2009; Papoulias et al., 2009). Phenolic content is considered a significant factor in breeders selection of a high antioxidant activity breeding line, and the phytochemical profile of various asparagus varieties provides information on the health-promoting potential of asparagus.
Free Amino Acid Composition
Free amino acids are important biologically active compounds, and some amino acids, as well as components of proteins and polypeptides, have proven to be essential for metabolism, growth, reproduction, and maintenance of immune function (López et al., 1996; Diana et al., 2014; Ito et al., 2017). The free amino acids present in asparagus seedlings are diverse and vary in content depending on the variety. Sixteen amino acids were analyzed in the seedlings of 23 varieties of asparagus (Table 2): alanine, arginine, asparagine, aspartic acid, γ-amino-n-butyric acid (GABA), glutamic acid, glycine, isoleucine, leucine, lysine, phenylalanine, proline, serine, threonine, tyrosine, and valine.
Table 2. Free amino acid compositions (μg⦁g-1 DW) of asparagus seedlings of 23 varieties
Table 2. Free amino acid compositions (μg⦁g-1 DW) of asparagus seedlings of 23 varieties (Continued)
yMeans with different letters in each column are significantly different (p ≤ 0.05) based on DMRT (n = 3).
The major amino acids of asparagus seedlings were proline, GABA, aspartic acid, and alanine. ‘ASM’ (9,000.8 µg·g-1 DW) contained the most amino acids. In descending order, ‘GUM’, ‘MOO’, ‘PUP’, ‘AVM’, and ‘ARE’ contained amino acid contents greater than 7,000 µg·g-1 DW. The lowest total amino acid content was 1,763 µg·g-1 DW (in ‘MAM’), which was 5 times lower than that observed in the ‘ASM’ variety. In particular, GABA and proline are reported to regulate plant responses to various stress conditions, such as inorganic element deficiency (Keutgen and Pawelzik, 2008). Notably, the ‘MAM’ variety showed the highest antioxidant activity and bioactive compound content but the lowest total amino acid content. There was no difference in the amino acid content of asparagus seedlings according to color (green or purple). The total amino acid content in the asparagus seedlings was lower than the amino acid content of the asparagus spears, which in fact contained more than four times the content observed in the seedlings containing the greatest amount of amino acids (‘ASM’). Studies have shown that the total amino acid content of asparagus spears is 55 mg·g-1 DW and that the amino acid content tends to increase with storage time at room temperature (King et al., 1990). In the UC-150 variety, 400 mg·g-1 DW or more total amino acids was observed, and the content varied depending on spear thickness, reaching a maximum when the diameter was intermediate (9–11 mm). In contrast, the amino acid content was lower in fine (≤8 mm) and extra thick (≥20 mm) spears. In addition, changes in the amino acid content with various methods of processing have been observed (López et al., 1996). Since asparagus seedlings are at an early stage of plant growth, it is thought that primary metabolites such as amino acids increase as spears grow.
GABA, one of the major amino acids present in asparagus seedlings, is classified as a minor amino acid in spears: There is a trace amount (<1 mg·g-1 DW) in spears but a large amount in seedlings (>200 mg·g-1 DW) (López et al., 1996). However, at the seedling stage, the ‘ASM’ variety exhibited the highest (9%) total amino acid content, and even ‘MAM’ exhibited a GABA content of approximately 16%. GABA is present in various plants, albeit in small amounts, such as spinach, potato, cabbage, asparagus, broccoli, apple, grape, cereal, and maize. In particular, germinated millets and legumes have been suggested to be a good source of GABA (Pradeep et al., 2011). GABA is a non-protein amino acid that is generated to some degree in the human body. In animals, it is present in high concentrations in the brain and plays a fundamental role in inhibitory neurotransmission in some pathways in the central nervous system and peripheral tissues (Sasaki et al., 2006; Diana et al., 2014). GABA has physiological functions that relieve restlessness, insomnia, and depression (Patil and Khan, 2011; Wu et al., 2014). The most important effect of GABA is blood pressure control, which has been reported in many studies. Blood pressure was reduced when rats and humans with hypertension were fed GABA-enriched foods (Hayakawa et al., 2004; Kajimoto et al., 2004).
Several amino acids are the main determinants of taste in fruits. For example, glycine, alanine, and proline provide sweetness, while leucine, isoleucine, and valine provide a bitter taste (Ito et al., 2017). Asparagus spears have a slightly sweet taste rather than a bitter taste. In this study, in asparagus seedlings, the level of amino acids conferring a sweet taste was higher than that of amino acids conferring a bitter taste. Thus, in asparagus, the alanine and proline contents increase, which increases sweetness, from the seedling stage to the spear stage. Another of the major amino acids present in asparagus is asparagine. Asparagine in plants is produced by three main pathways: The first pathway is mediated by asparagine-oxoacid transaminase, the second is mediated by cyanide and cysteine incorporated into p-cyanoalanine, and the third involves production from aspartic acid using glutamine as a nitrogen donor. In asparagus, asparagine is mainly produced by the third route (Sieciechowicz et al., 1988; Lea et al., 2007). Asparagine produced by the third route is produced by aspartic acid. There was a large amount of aspartic acid in the asparagus seedlings used in this study. Therefore, a large amount of asparagine must have been produced. Among the asparagus varieties, ‘ASM’ contained the most aspartic acid and asparagine at the seedling stage. Cysteine, the second route for the synthesis of asparagine, is also used for asparagine synthesis. In particular, the asparagine content of ‘ASM’, ‘ATS’, and ‘PAP’, which are high in cysteine as well as aspartic acid, may be increased (data not shown).
Conclusion
The results of this study indicate that there are various types of bioactive compounds in asparagus seedlings. Although there have been many studies using spears, few studies have considered the existence of various kinds of active compounds in asparagus seedlings. The useful substances present in asparagus seedlings are diverse and highly dependent on the variety. However, there was a significant difference between high antioxidant activity and bioactive compound content and high free amino acid content. The varieties (approximately six) with high antioxidant activity and high bioactive compound content were similar to each other, whereas the varieties with higher free amino acid content were completely different from each other. Notably, the free amino acid content of ‘MAM’ was the lowest, even though this variety had the highest antioxidant activity and bioactive compound content. In contrast, ‘ASM’ showed the highest free amino acid content, while its antioxidant capacity, rutin, and nicotiflorin were the lowest. Studies using asparagus seedlings cannot fully determine the useful compound content in asparagus spears, but they do increase the potential for producing asparagus varieties that contain many bioactive compounds helpful to producers and breeders.
Supplementary Material
Supplementary materials are available at Horticultural Science and Technology website (https://www.hst-j.org).
- HORT_20200028_Table_1s.pdf
Total phenolic content and total flavonoid content in asparagus seedlings of 23 varieties
- HORT_20200028_Table_2s.pdf
Antioxidant activities in asparagus seedlings of 23 varieties
- HORT_20200028_Table_3s.pdf
Flavonoid contents of 23 varieties of asparagus seedlings
- HORT_20200028_Fig_1s.pdf
HPLC chromatogram of rutin and nicotiflorin in asparagus seedlings.
- HORT_20200028_Fig_2s.pdf
HPLC chromatogram of amino acids in asparagus seedlings of ‘Atlas’.
- HORT_20200028_Fig_3s.pdf
HPLC chromatogram of amino acids in asparagus seedlings of ‘UC157’.